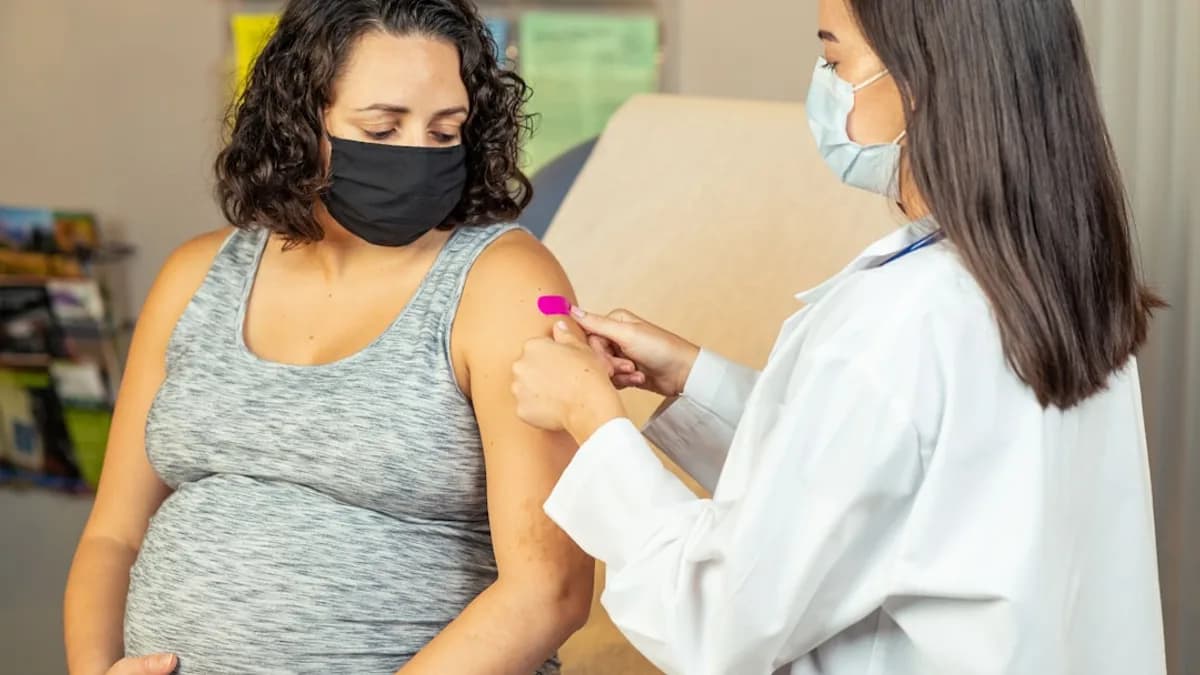
Introduction: Understanding RSV and Its Impact on Infants
Respiratory Syncytial Virus (RSV) is a highly contagious virus that infects the respiratory tract, affecting people of all ages but posing the greatest risk to infants and young children. Nearly every child will contract RSV by their second birthday, but for newborns, especially those under six months old, the infection can be severe. RSV is a leading cause of bronchiolitis and pneumonia in infants, often resulting in hospitalization and, in rare cases, life-threatening complications.
Each year, RSV leads to thousands of hospital admissions for babies worldwide, straining families and healthcare systems alike. The vulnerability of young infants, whose immune systems are still developing, makes preventive strategies crucial. For decades, efforts to protect this at-risk population have focused on hygiene, limiting exposure, and, more recently, the development of targeted vaccines. The emergence of a safe and effective maternal RSV vaccine could represent a significant breakthrough in safeguarding newborns during their most vulnerable early months.
What Is the New Pregnancy Vaccine Against RSV?
The new vaccine targeting RSV is specifically designed for pregnant women with the goal of protecting both mother and baby. Administered during the late stages of pregnancy—typically in the third trimester—the vaccine prompts the mother's immune system to produce protective antibodies against RSV. These antibodies are then naturally transferred to the fetus through the placenta, equipping the newborn with ready-made defenses against the virus from birth.
Unlike previous approaches, which have focused on vaccinating infants directly or offering monoclonal antibody treatments, this maternal vaccine leverages the well-established principle of passive immunity. By delivering the vaccine before birth, the intervention offers immediate protection during the critical first months of life when infants are most susceptible to severe RSV outcomes.
The timeline for administration is carefully selected to maximize antibody transfer without interfering with labor and delivery. As regulatory agencies review safety and efficacy data, recommendations are being developed to guide healthcare providers on optimal timing and eligibility for expectant mothers.
Evidence from Recent Studies: Effectiveness of the Vaccine
Compelling evidence supporting the effectiveness of the RSV vaccine for pregnant women comes from a comprehensive systematic review and meta-analysis published in Nature. The analysis, which pooled data from multiple randomized controlled trials, found that maternal vaccination against RSV led to a significant reduction in serious infant outcomes—including an 80% decrease in hospital admissions for RSV among newborns [Source: Source].
This finding is particularly noteworthy given the high risk of hospitalization due to RSV in the first months of life. By preventing the most severe cases, the vaccine not only safeguards infants but also alleviates the strain on pediatric healthcare services.
Regulatory progress is also underway. In the United States, the FDA’s independent advisory committee recently voted in favor of approving an RSV vaccine for use in pregnancy, citing robust evidence of safety and efficacy [Source: Source]. This support paves the way for broader accessibility and incorporation into routine maternal immunization practices.
Other studies have echoed these positive results. For example, coverage by Inside Precision Medicine highlights that vaccination in late pregnancy substantially reduces the risk of RSV-related hospitalizations for infants, reinforcing the real-world impact of this preventive strategy [Source: Source]. As more countries evaluate the evidence, international guidelines for maternal RSV vaccination are expected to follow.
How Maternal Vaccination Protects Newborns
The protective effect of maternal RSV vaccination hinges on the transfer of antibodies from mother to fetus during pregnancy. When a pregnant woman receives the vaccine, her immune system generates high levels of RSV-specific antibodies. These antibodies cross the placenta, reaching the developing baby and providing passive immunity that persists through the first critical months of life.
This maternal-infant antibody transfer is a well-established mechanism, already employed in vaccination strategies for diseases like influenza and pertussis. In the case of RSV, the transferred antibodies significantly lower the risk of severe infection and hospitalization during the period when infants are most vulnerable and too young to receive vaccines themselves.
The duration of this protection typically spans the initial months after birth, which aligns with the period of highest RSV risk. In addition to reducing hospitalizations, studies suggest that maternal vaccination may also improve broader perinatal outcomes by decreasing complications associated with severe respiratory infections [Source: Source]. However, ongoing research is examining the optimal balance between timing, dosage, and long-term benefits for both mothers and infants.
Implications for Public Health and Future Directions
The introduction of a maternal RSV vaccine has transformative potential for public health. By preventing up to 80% of RSV-related hospital admissions among infants, widespread vaccination could dramatically reduce the disease burden on families and healthcare systems. Fewer hospitalizations mean less emotional stress for parents, lower healthcare costs, and reduced risk of long-term respiratory complications in children [Source: Source].
However, achieving these benefits depends on high vaccine uptake and effective communication with pregnant women. Misinformation, vaccine hesitancy, and logistical challenges in prenatal care can hinder adoption. Public health campaigns and clear guidance from healthcare providers will be essential to ensure that expectant mothers understand the risks of RSV and the protective value of maternal immunization.
Looking ahead, ongoing research aims to refine RSV vaccines for even greater efficacy and safety. Scientists are also exploring additional strategies, such as combining RSV vaccination with other maternal vaccines or developing long-acting monoclonal antibodies for infants. As more data emerge, policymakers and clinicians will need to adapt recommendations to maximize protection for the youngest and most vulnerable members of society.
Conclusion: The Promise of Maternal RSV Vaccination
The development and validation of a maternal RSV vaccine mark a critical advance in the fight against severe respiratory infections in infants. With evidence showing a dramatic reduction in hospital admissions, maternal immunization stands out as a powerful preventive tool. As regulatory approvals progress and public awareness grows, informed discussions between healthcare providers and expectant mothers will be key to realizing the full potential of this life-saving intervention. By embracing maternal RSV vaccination, we can take an important step toward safer, healthier beginnings for newborns everywhere.